
FVIIa analogue
Category
FVIIa
ID
NNC0078-0007
Amount per vial
2.2 mg
Vatreptacog alfa (see the Persson E et al. reference listed in the reference section below) is a variant of human coagulation factor VIIa (hFVIIa), also known as “rFVIIa analog NN1731” or “FVIIaDVQ”. It is a two-chain recombinant protein produced in mammalian cells. The heavy and light chains are connected via a disulfide bond. The LC features an N-terminal Gla-domain containing multiple gamma-carboxy glutaminyl residues as well as two EGF-like domains. The catalytic domain is part of the HC. Compared to hFVIIa, vatreptacog alfa contains three amino acid changes, V158D, E296V, and M298Q. These substitutions result in considerable enhancement of the intrinsic (tissue factor-independent) activity. The enhanced activity of vatreptacog alfa has been demonstrated in numerous in vitro systems and in vivo model mimicking hemophilia. Vatreptacog alfa has been investigated in phase 1, 2, and 3 clinical trials. Vatreptacog alfa was discontinued after phase 3 clinical trial due to anti-drug antibodies (Mahlangu J N et al. et al. - reference section) in a few patients. A post hoc assessment of the immunogenicity of vatreptacog alfa was subsequently performed in collaboration with FDA (see the Lamberth K et al. reference listed in the reference section below).
Category
FVIIa
ID
NNC0078-0007
Amount per vial
2.2 mg
| Parameter | NNC0078-0007 |
| pI | 5.6 |
| MW average | 50 kDa |
| MW LC | 20 kDa |
| MW HC | 30 kDa |
| A280 (1 mg/ml) | 1.37 |
| Glycosylations | Asn145 and Asn322 (complex biatennary) |
| Max thrombin generation rate | 3-10 times higher than rhFVIIa (see fourth reference below) |
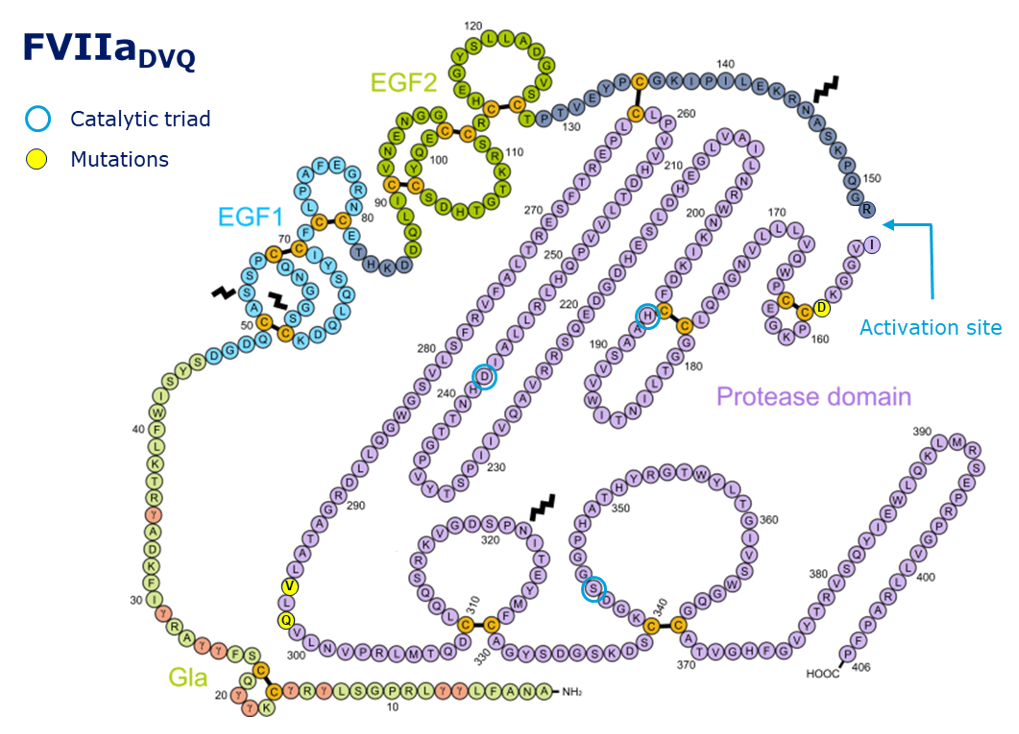
Figure 1
2D structure of vatreptacoq alfa with domains, post-translational modifications, and special residues indicated.
| Properties | NNC0078-0007 |
| Content | 2.2 mg/vial |
| HMWP | 2.4% |
| Total impurities | 9.7% |
| Oxidized forms | 2.7% |
| Heavy chain degradation | 4.3% |
| Gamma-carboxylation | Complies |
| N-terminal sequence | Complies |
| Specific activity | 1085 U/ug |
| Bacterial Endotoxin | <0.10 U/mg |